https://doi.org/10.1016/j.drudis.2026.104678 Free download of the article using this link until June 16: https://authors.elsevier.com/a/1n0uP4r9Rkz1l6
Highlights
-
New four-pillar framework accelerates deorphanization of dark GPCR targets. (77 chars)
-
GPR149 structural analysis identifies non-canonical ERY and DPxxF motifs. (76 chars)
-
Path-agnostic screening bypasses traditional Gi/o signaling limitations. (75 chars)
-
Integrated CNS and metabolic mapping reveals GPR149′s dual-domain value. (76 chars)
-
Blueprint provides high-resolution de-risking for first-in-class assets. (74 chars)
High failure rates in drug development for central nervous system (CNS) and metabolic diseases frequently stem from a lack of knowledge about their selected drug targets. With unknown ligand chemistry, orphan G-protein-coupled receptors (GPCRs) represent high-risk, but high-potential, high-reward opportunities for pharmaceutical development. Here, I describe a framework for de-risking such targets using GPR149 as a prototype. The Four-Pillar Framework, combining high-throughput screening, cryo-electron microscopy (EM), artificial intelligence (AI)-driven chemistry, and parallel circuit validation, unexpectedly revealed the dual metabolic (weight loss) and CNS applications of GPR149. In the process, a seemingly intractable orphan receptor has become a development asset with blockbuster potential. This methodology offers a reproducible template for exploring the ‘dark GPCRome’, particularly for disorders in which metabolic dysfunction and CNS comorbidities co-present in real-world patient populations.
Graphical abstract
Keywords
orphan GPCR
GPR149
deorphanization
target validation
dual-domain therapeutics
drug discovery pipeline
cryo-EM
AI-driven chemistry
circuit-level pharmacology
incremental risk mitigation
Introduction: prioritizing and de-risking the dark GPCRome
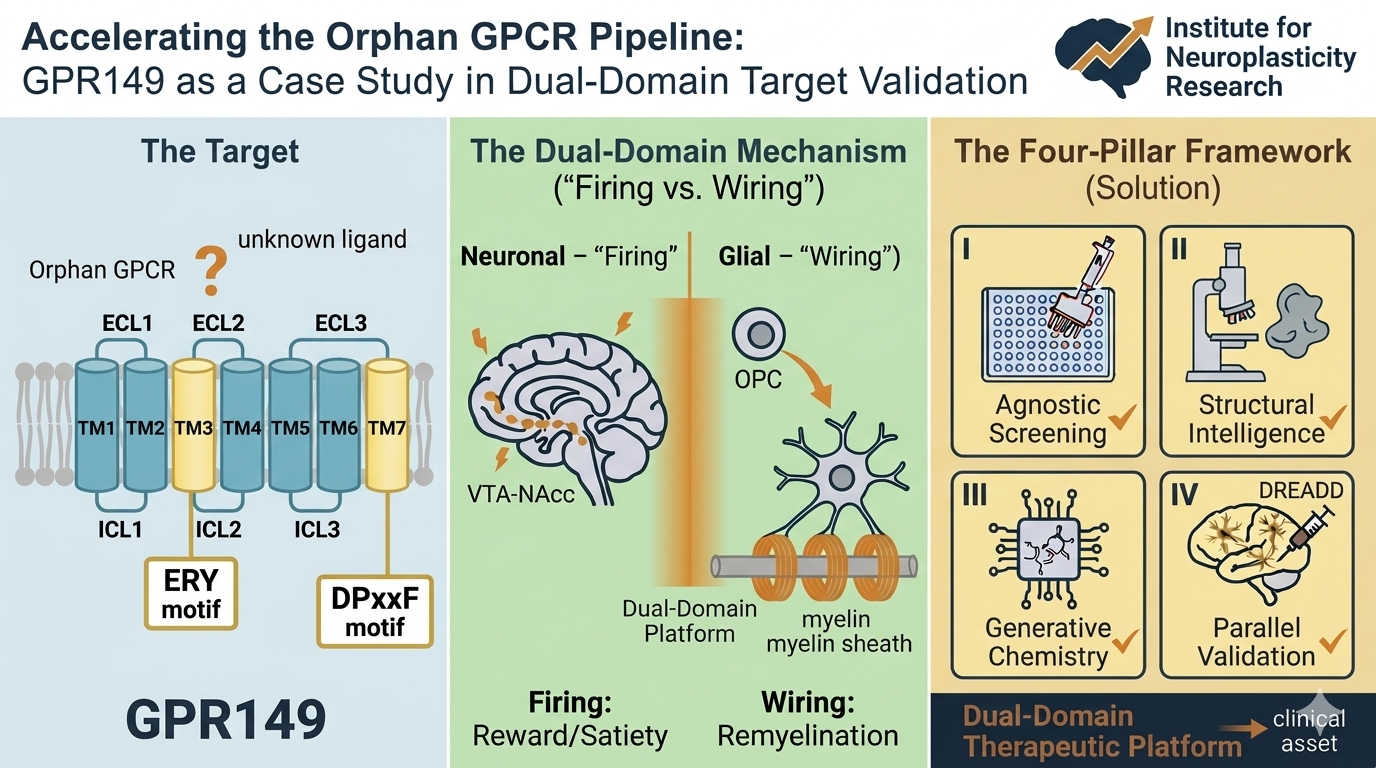
The ‘dark GPCRome’ represents one of the most significant untapped frontiers in modern drug discovery. Although GPCRs remain the most successful class of drug targets, accounting for ∼35% of all US Food and Drug Administration (FDA)-approved therapeutics and nearly 60% of current prescriptions, most of this success is concentrated within a well-trodden subset of this superfamily.(p1),(p2) Most approved agents target the Class A (rhodopsin-like) subfamily, characterized by the seven-transmembrane helix architecture and highly conserved signaling motifs, such as DRY, CWxP, and NPxxY.(p3) However, a modern drug discovery lens necessitates moving beyond these established targets to de-risk ‘dark’ receptors that deviate from these canonical sequences, where structural and functional gaps have historically stalled development.
GPR149 epitomizes this non-canonical challenge. Although phylogenetically classified within the rhodopsin-like subfamily, GPR149 lacks the crucial charged residues of the hallmark Asp-Arg-Tyr (DRY) motif, featuring instead a divergent ERY triplet.(p4) This specific substitution at the 3.50 position (Ballesteros–Weinstein numbering) is not merely a sequence variation; it likely dictates high constitutive activity and unconventional G-protein coupling of the receptor.
Despite being cloned nearly a quarter-century ago (initially as PGR10), GPR149 remains a classic orphan, trapped in the ‘valley of death’ between academic phenotypic discovery and commercial R&D advancement.(p5)
The productivity paradox: Eroom’s Law
This stagnation is not merely a function of difficult biology. It reflects a well-documented phenomenon known as ‘Eroom’s Law’ (‘Moore’s Law’ spelled backward). First articulated during the early 2010s, Eroom’s Law describes the observation that the number of new drugs approved per billion dollars spent on pharmaceutical R&D has halved approximately every 9 years since 1950. This trend persists despite, or perhaps because of, technological advances in screening, computing, and molecular biology. The drivers include what has been termed the ‘better than the Beatles’ problem (new drugs must compete against an ever-improving catalog of effective generics), increasing regulatory caution, diminishing returns from brute-force screening approaches, and the tendency to simply allocate more resources to failing strategies rather than rethinking the underlying logic of discovery.(p1),(p2),(p3)
Orphan GPCRs sit at the epicenter of this challenge. They offer high-reward opportunities not simply because they are unexplored, but because their anatomical expression patterns, strategically enriched in hypothalamic feeding circuits, mesolimbic reward pathways, and glial populations governing myelination, position them as master regulators of physiology with direct therapeutic relevance. GPR149 exemplifies this logic: its localization to the arcuate hypothalamus and nucleus accumbens, coupled with validated roles in energy homeostasis and oligodendrocyte progenitor cell (OPC) differentiation, transforms an orphan receptor from a biological mystery into a strategic asset. Yet, this promise carries correspondingly high risk because of non-canonical signaling motifs, unknown ligand chemistry, and uncertain clinical translatability. Therefore, a paradigm shift is required: one that treats deorphanization not as a sequential hunt for a ligand but as an integrated, parallelized de-risking campaign.
A paradigm shift in target validation
Historically, deorphanization has been a step-by-step process hampered by long timelines and high failure rates. Final deorphanization occurred, in part, due to luck. Today, a paradigm shift is possible through the combination of disruptive technologies: multiplexed functional assays, AI-driven de novo design, and cryo-electron microscopy (cryo-EM). By enabling near-atomic-resolution imaging of fragile GPCR complexes in their native states without the need for crystallization, cryo-EM, coupled with generative AI, allows researchers to visualize dynamic biological mechanisms in action. Together, these tools offer the potential to methodically de-risk the entire biology of a target simultaneously, rather than simply hunting for a ligand.
GPR149: a prototype for dual-domain success
GPR149 is an ideal model because of its precise anatomical localization in circuits governing reward and energy homeostasis, specifically the nucleus accumbens in rodents (and potentially in humans) and the arcuate hypothalamus in humans.(p5),(p6) Phenotypic findings in Gpr149-null animals, which exhibit improved insulin sensitivity and altered energy homeostasis, provide the initial metabolic impetus.(p8) However, the clinical relevance of GPR149 is anchored by high-resolution human transcriptomic data, which confirm its expression in hypothalamic energy-management nodes, providing the species-specific justification required for an industrial de-risking campaign.(p5)
Beyond metabolism, GPR149 is a validated regulator of OPC function, suggesting a parallel role in CNS remyelination.(p9) In pathological states, GPR149 signaling contributes to the inhibitory environment that prevents OPCs from maturing into functional, myelin-producing oligodendrocytes, a hallmark of chronic demyelinating diseases such as multiple sclerosis (MS). This positions GPR149 not merely as a metabolic regulator but as a true ‘neuro-metabolic-glial platform’, a single target addressing both the synaptic ‘firing’ of reward circuitry and the structural ‘wiring’ of white matter integrity.Published GPR149 article




 Posted in
Posted in