𝐅𝐨𝐜𝐮𝐬𝐢𝐧𝐠 𝐨𝐧 𝐛𝐞𝐭𝐭𝐞𝐫 𝐚𝐥𝐭𝐞𝐫𝐧𝐚𝐭𝐢𝐯𝐞𝐬 𝐭𝐨 𝐬𝐡𝐢𝐩𝐩𝐢𝐧𝐠 𝐨𝐟𝐟 𝐞𝐥𝐝𝐞𝐫𝐥𝐲 𝐩𝐚𝐫𝐞𝐧𝐭𝐬 𝐭𝐨 𝐧𝐮𝐫𝐬𝐢𝐧𝐠 𝐡𝐨𝐦𝐞𝐬. Moira Welsh’s HAPPILY EVER OLDER is a hopeful investigative work that serves as a “blueprint for change” in long-term care . Instead of focusing on common horror stories, Welsh travels across North America and Europe to find facilities that have successfully replaced institutional “warehousing” with vibrant […]
Archive for the ‘Regulatory Medical Writing’ Category
𝐅𝐨𝐜𝐮𝐬𝐢𝐧𝐠 𝐨𝐧 𝐛𝐞𝐭𝐭𝐞𝐫 𝐚𝐥𝐭𝐞𝐫𝐧𝐚𝐭𝐢𝐯𝐞𝐬 𝐭𝐨 𝐬𝐡𝐢𝐩𝐩𝐢𝐧𝐠 𝐨𝐟𝐟 𝐞𝐥𝐝𝐞𝐫𝐥𝐲 𝐩𝐚𝐫𝐞𝐧𝐭𝐬 𝐭𝐨 𝐧𝐮𝐫𝐬𝐢𝐧𝐠 𝐡𝐨𝐦𝐞𝐬
𝐅𝐨𝐜𝐮𝐬 𝐨𝐧 𝐄𝐦𝐩𝐨𝐰𝐞𝐫𝐦𝐞𝐧𝐭 & 𝐄𝐝𝐮𝐜𝐚𝐭𝐢𝐨𝐧: 𝐀𝐫𝐞 𝐲𝐨𝐮 𝐛𝐮𝐢𝐥𝐝𝐢𝐧𝐠 𝐲𝐨𝐮𝐫 𝐛𝐫𝐚𝐢𝐧 𝐮𝐩, 𝐨𝐫 𝐚𝐫𝐞 𝐲𝐨𝐮 𝐥𝐞𝐭𝐭𝐢𝐧𝐠 𝐢𝐭 𝐛𝐫𝐞𝐚𝐤 𝐝𝐨𝐰𝐧?

𝐅𝐨𝐜𝐮𝐬 𝐨𝐧 𝐄𝐦𝐩𝐨𝐰𝐞𝐫𝐦𝐞𝐧𝐭 & 𝐄𝐝𝐮𝐜𝐚𝐭𝐢𝐨𝐧: 𝐀𝐫𝐞 𝐲𝐨𝐮 𝐛𝐮𝐢𝐥𝐝𝐢𝐧𝐠 𝐲𝐨𝐮𝐫 𝐛𝐫𝐚𝐢𝐧 𝐮𝐩, 𝐨𝐫 𝐚𝐫𝐞 𝐲𝐨𝐮 𝐥𝐞𝐭𝐭𝐢𝐧𝐠 𝐢𝐭 𝐛𝐫𝐞𝐚𝐤 𝐝𝐨𝐰𝐧? The concept of Cognitive Reserve is the single most empowering framework for understanding how we age. “Cognitive Reserve” is the buffer your brain builds—a dynamic network of strong neural connections that protects your ability to think, remember, […]
Authority, Long-term Reach, and “Save” Signals
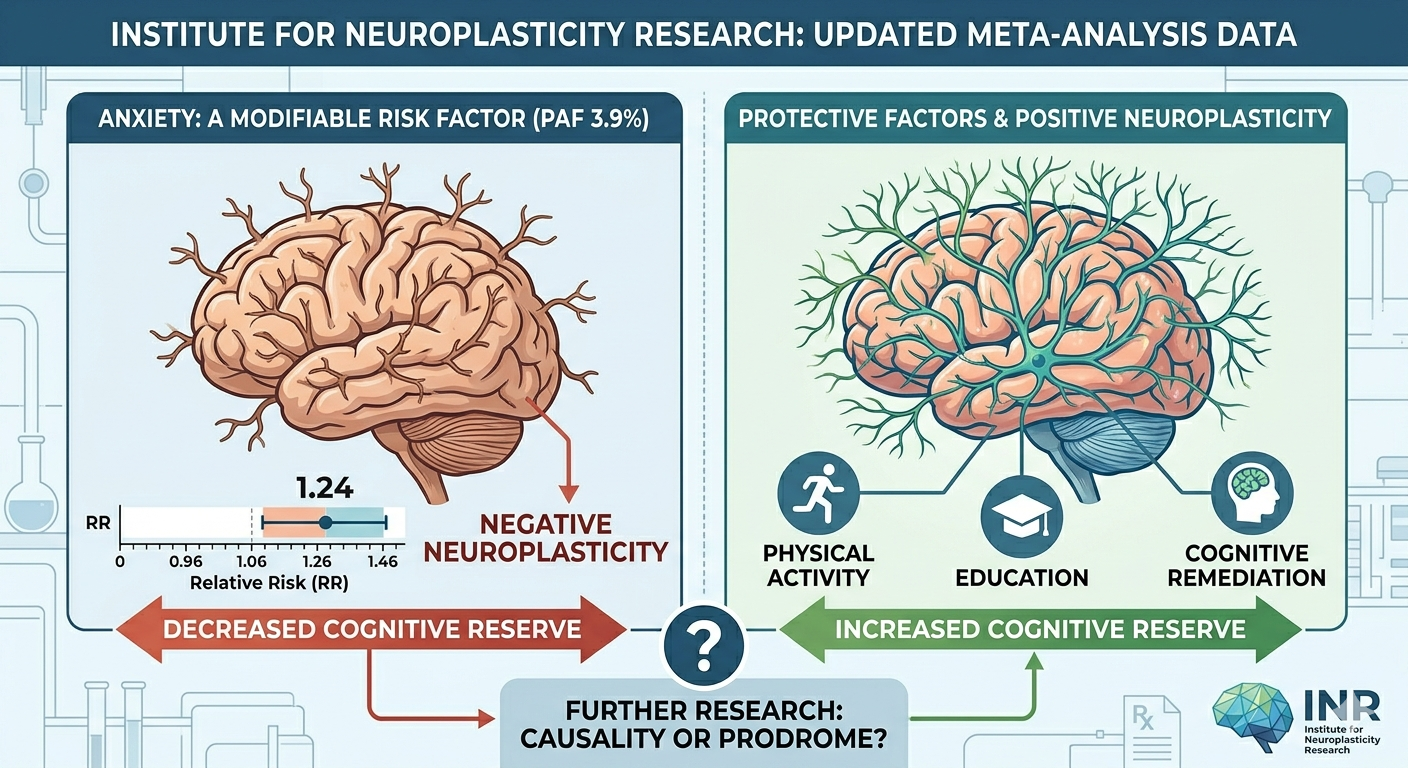
New evidence provides a stronger link between chronic anxiety and future neurocognitive decline, demanding a re-evaluation of how we approach dementia prevention in clinical practice. A major updated meta-analysis, now available in the Journal of Clinical Medicine, confirms that anxiety is significantly associated with an increased risk of all-cause dementia. This extensive study, spanning nine […]
SuperAgers Produce More Neuroblasts
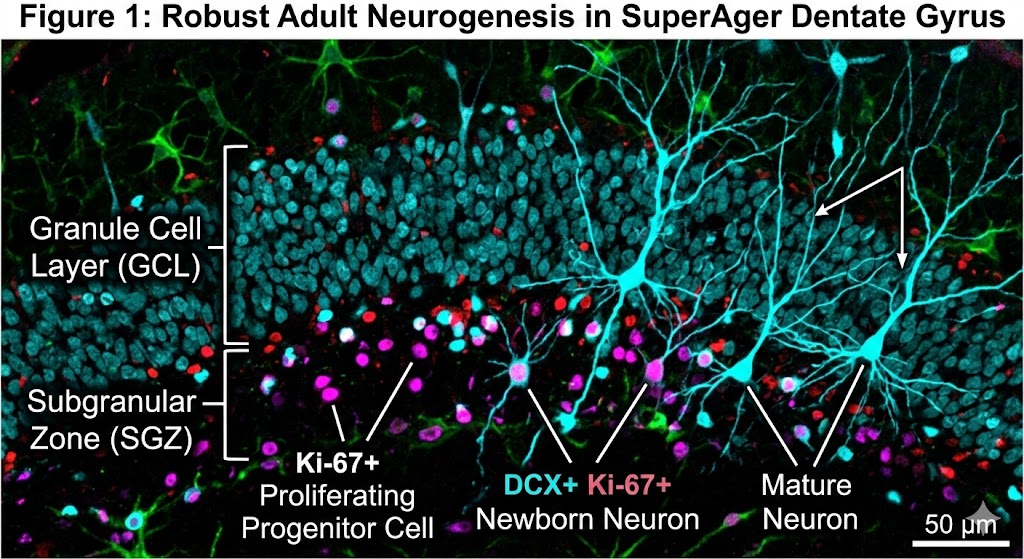
The dogma that neurogenesis slows to a trickle in the aging human brain has been significantly challenged this week. A groundbreaking study published in Nature (February 25, 2026), led by scientists at the University of Illinois Chicago and the Northwestern University SuperAger Program, reveals that SuperAgers produce neuroblasts at rates far higher than their age-matched […]
𝐘𝐨𝐮 𝐀𝐫𝐞 𝐭𝐡𝐞 𝐀𝐫𝐜𝐡𝐢𝐭𝐞𝐜𝐭: 𝐓𝐡𝐞 𝐏𝐨𝐰𝐞𝐫 𝐨𝐟 𝐏𝐨𝐬𝐢𝐭𝐢𝐯𝐞 𝐍𝐞𝐮𝐫𝐨𝐩𝐥𝐚𝐬𝐭𝐢𝐜𝐢𝐭𝐲

𝐒𝐭𝐨𝐩 𝐭𝐡𝐢𝐧𝐤𝐢𝐧𝐠 𝐨𝐟 𝐲𝐨𝐮𝐫 𝐛𝐫𝐚𝐢𝐧 𝐚𝐬 𝐚 𝐬𝐭𝐚𝐭𝐢𝐜 𝐨𝐫𝐠𝐚𝐧 𝐭𝐡𝐚𝐭 𝐨𝐧𝐥𝐲 𝐝𝐞𝐜𝐥𝐢𝐧𝐞𝐬 𝐰𝐢𝐭𝐡 𝐚𝐠𝐞. 𝐈𝐧 𝟐𝟎𝟐𝟔, 𝐰𝐞 𝐡𝐚𝐯𝐞 𝐭𝐡𝐞 𝐜𝐨𝐦𝐩𝐮𝐭𝐚𝐭𝐢𝐨𝐧𝐚𝐥 𝐩𝐫𝐨𝐨𝐟 𝐭𝐡𝐚𝐭 𝐲𝐨𝐮 𝐚𝐫𝐞 𝐭𝐡𝐞 𝐚𝐫𝐜𝐡𝐢𝐭𝐞𝐜𝐭 𝐨𝐟 𝐲𝐨𝐮𝐫 𝐨𝐰𝐧 𝐠𝐫𝐞𝐲 𝐦𝐚𝐭𝐭𝐞𝐫. 𝐖𝐞 𝐜𝐚𝐥𝐥 𝐭𝐡𝐢𝐬 𝐏𝐨𝐬𝐢𝐭𝐢𝐯𝐞 𝐍𝐞𝐮𝐫𝐨𝐩𝐥𝐚𝐬𝐭𝐢𝐜𝐢𝐭𝐲, 𝐚𝐧𝐝 𝐢𝐭 𝐢𝐬 𝐭𝐡𝐞 𝐦𝐨𝐬𝐭 𝐞𝐦𝐩𝐨𝐰𝐞𝐫𝐢𝐧𝐠 𝐬𝐜𝐢𝐞𝐧𝐭𝐢𝐟𝐢𝐜 𝐫𝐞𝐚𝐥𝐢𝐭𝐲 𝐨𝐟 𝐨𝐮𝐫 𝐭𝐢𝐦𝐞. 𝐘𝐨𝐮𝐫 𝐛𝐫𝐚𝐢𝐧 𝐢𝐬 𝐚 𝐝𝐲𝐧𝐚𝐦𝐢𝐜, 𝐟𝐥𝐮𝐢𝐝 𝐜𝐢𝐫𝐜𝐮𝐢𝐭 𝐭𝐡𝐚𝐭 […]
Beyond the BBB: Mapping the 2026 Milestone Year for Neuroscience
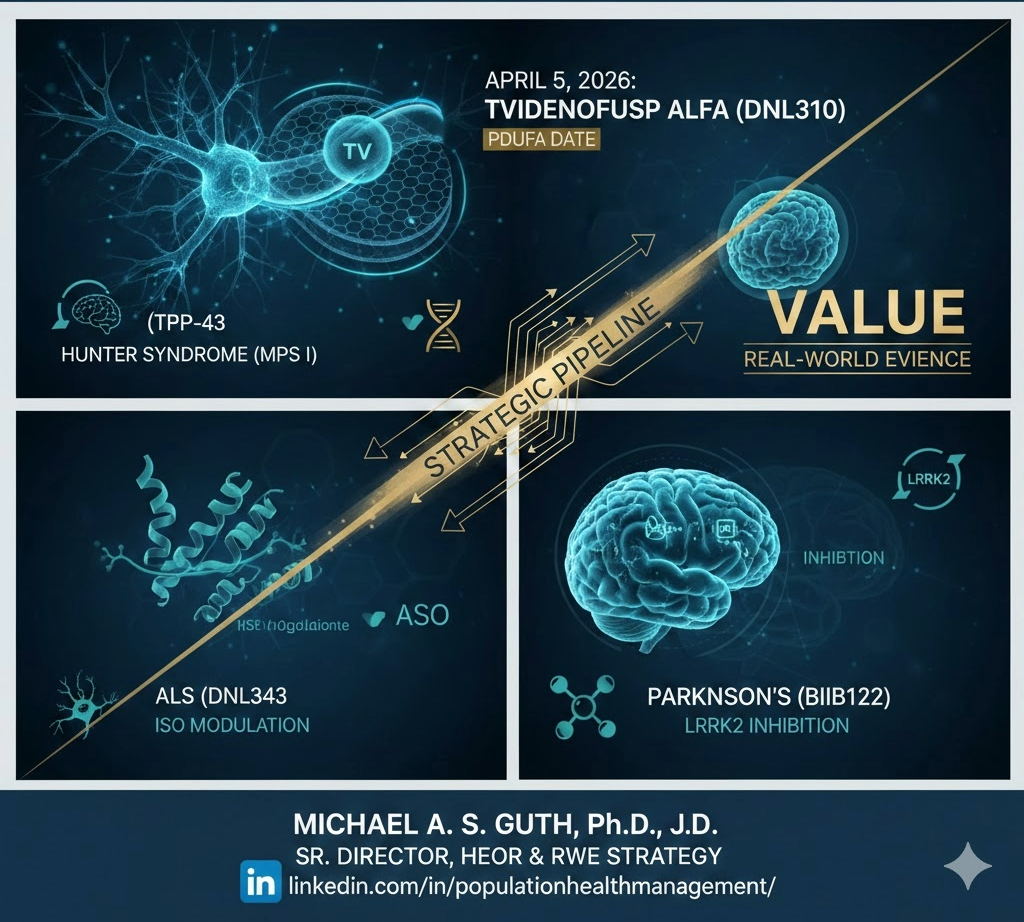
Beyond the BBB: Mapping the 2026 Milestone Year for Neuroscience. As we move through Q1, all eyes in the neuroscience community are on April 5, 2026—the PDUFA target action date for tividenofusp alfa (DNL310). If approved, tividenofusp alfa will be a watershed moment: the first commercial validation of a Transport Vehicle (TV) enabled enzyme replacement […]
𝐖𝐡𝐲 𝐃𝐍𝐀 𝐎𝐫𝐢𝐠𝐚𝐦𝐢 𝐢𝐬 𝐭𝐡𝐞 𝐒𝐞𝐜𝐫𝐞𝐭 𝐭𝐨 𝐚 “𝐒𝐢𝐥𝐞𝐧𝐭” 𝐕𝐚𝐜𝐜𝐢𝐧𝐞 𝐒𝐜𝐚𝐟𝐟𝐨𝐥𝐝 𝐈𝐧 𝐯𝐚𝐜𝐜𝐢𝐧𝐞 𝐝𝐞𝐬𝐢𝐠𝐧,
𝐖𝐡𝐲 𝐃𝐍𝐀 𝐎𝐫𝐢𝐠𝐚𝐦𝐢 𝐢𝐬 𝐭𝐡𝐞 𝐒𝐞𝐜𝐫𝐞𝐭 𝐭𝐨 𝐚 “𝐒𝐢𝐥𝐞𝐧𝐭” 𝐕𝐚𝐜𝐜𝐢𝐧𝐞 𝐒𝐜𝐚𝐟𝐟𝐨𝐥𝐝 𝐈𝐧 𝐯𝐚𝐜𝐜𝐢𝐧𝐞 𝐝𝐞𝐬𝐢𝐠𝐧, “𝐨𝐟𝐟-𝐭𝐚𝐫𝐠𝐞𝐭” 𝐫𝐞𝐬𝐩𝐨𝐧𝐬𝐞𝐬 𝐚𝐫𝐞 𝐚 𝐩𝐞𝐫𝐬𝐢𝐬𝐭𝐞𝐧𝐭 𝐡𝐮𝐫𝐝𝐥𝐞. 𝐖𝐡𝐞𝐧 𝐰𝐞 𝐮𝐬𝐞 𝐩𝐫𝐨𝐭𝐞𝐢𝐧-𝐛𝐚𝐬𝐞𝐝 𝐩𝐚𝐫𝐭𝐢𝐜𝐥𝐞𝐬 𝐭𝐨 𝐝𝐞𝐥𝐢𝐯𝐞𝐫 𝐚𝐧 𝐚𝐧𝐭𝐢𝐠𝐞𝐧, 𝐭𝐡𝐞 𝐢𝐦𝐦𝐮𝐧𝐞 𝐬𝐲𝐬𝐭𝐞𝐦 𝐨𝐟𝐭𝐞𝐧 𝐠𝐞𝐭𝐬 𝐝𝐢𝐬𝐭𝐫𝐚𝐜𝐭𝐞𝐝 𝐚𝐧𝐝 𝐚𝐭𝐭𝐚𝐜𝐤𝐬 𝐭𝐡𝐞 𝐝𝐞𝐥𝐢𝐯𝐞𝐫𝐲 𝐯𝐞𝐡𝐢𝐜𝐥𝐞 𝐢𝐭𝐬𝐞𝐥𝐟 𝐢𝐧𝐬𝐭𝐞𝐚𝐝 𝐨𝐟 𝐭𝐡𝐞 𝐯𝐢𝐫𝐮𝐬. 𝐍𝐞𝐰 𝐫𝐞𝐬𝐞𝐚𝐫𝐜𝐡 𝐟𝐫𝐨𝐦 𝐌𝐈𝐓 𝐚𝐧𝐝 𝐒𝐜𝐫𝐢𝐩𝐩𝐬 𝐑𝐞𝐬𝐞𝐚𝐫𝐜𝐡, 𝐩𝐮𝐛𝐥𝐢𝐬𝐡𝐞𝐝 𝐢𝐧 𝐒𝐜𝐢𝐞𝐧𝐜𝐞, […]
𝗡𝗜𝗖𝗘 𝗥𝗲𝗰𝗼𝗺𝗺𝗲𝗻𝗱𝘀 𝗗𝘂𝗽𝗶𝗹𝘂𝗺𝗮𝗯 𝗳𝗼𝗿 𝗘𝗼𝘀𝗶𝗻𝗼𝗽𝗵𝗶𝗹𝗶𝗰 𝗖𝗢𝗣𝗗: 𝗔 𝗗𝗲𝗲𝗽 𝗗𝗶𝘃𝗲 𝗶𝗻𝘁𝗼 𝘁𝗵𝗲 𝗘𝘃𝗶𝗱𝗲𝗻𝗰𝗲 𝗮𝗻𝗱 𝗘𝗰𝗼𝗻𝗼𝗺𝗶𝗰 𝗖𝗮𝘀𝗲
𝗡𝗜𝗖𝗘 𝗥𝗲𝗰𝗼𝗺𝗺𝗲𝗻𝗱𝘀 𝗗𝘂𝗽𝗶𝗹𝘂𝗺𝗮𝗯 𝗳𝗼𝗿 𝗘𝗼𝘀𝗶𝗻𝗼𝗽𝗵𝗶𝗹𝗶𝗰 𝗖𝗢𝗣𝗗: 𝗔 𝗗𝗲𝗲𝗽 𝗗𝗶𝘃𝗲 𝗶𝗻𝘁𝗼 𝘁𝗵𝗲 𝗘𝘃𝗶𝗱𝗲𝗻𝗰𝗲 𝗮𝗻𝗱 𝗘𝗰𝗼𝗻𝗼𝗺𝗶𝗰 𝗖𝗮𝘀𝗲 𝗧𝗵𝗲 𝗨𝗞’𝘀 𝗡𝗮𝘁𝗶𝗼𝗻𝗮𝗹 𝗜𝗻𝘀𝘁𝗶𝘁𝘂𝘁𝗲 𝗳𝗼𝗿 𝗛𝗲𝗮𝗹𝘁𝗵 𝗮𝗻𝗱 𝗖𝗮𝗿𝗲 𝗘𝘅𝗰𝗲𝗹𝗹𝗲𝗻𝗰𝗲 (𝗡𝗜𝗖𝗘) 𝗵𝗮𝘀 𝗶𝘀𝘀𝘂𝗲𝗱 𝗳𝗶𝗻𝗮𝗹 𝗱𝗿𝗮𝗳𝘁 𝗴𝘂𝗶𝗱𝗮𝗻𝗰𝗲 𝗿𝗲𝗰𝗼𝗺𝗺𝗲𝗻𝗱𝗶𝗻𝗴 𝗱𝘂𝗽𝗶𝗹𝘂𝗺𝗮𝗯 𝗮𝘀 𝗮𝗻 𝗮𝗱𝗱-𝗼𝗻 𝗺𝗮𝗶𝗻𝘁𝗲𝗻𝗮𝗻𝗰𝗲 𝘁𝗵𝗲𝗿𝗮𝗽𝘆 𝗳𝗼𝗿 𝗮 𝘀𝗽𝗲𝗰𝗶𝗳𝗶𝗰, 𝗵𝗶𝗴𝗵-𝗻𝗲𝗲𝗱 𝗖𝗢𝗣𝗗 𝗽𝗼𝗽𝘂𝗹𝗮𝘁𝗶𝗼𝗻. 𝗧𝗵𝗶𝘀 𝗺𝗮𝗿𝗸𝘀 𝗮 𝗽𝗶𝘃𝗼𝘁𝗮𝗹 𝘀𝗵𝗶𝗳𝘁 𝘁𝗼𝘄𝗮𝗿𝗱𝘀 𝗽𝗿𝗲𝗰𝗶𝘀𝗶𝗼𝗻 𝗺𝗲𝗱𝗶𝗰𝗶𝗻𝗲 𝗶𝗻 𝗰𝗵𝗿𝗼𝗻𝗶𝗰 𝗿𝗲𝘀𝗽𝗶𝗿𝗮𝘁𝗼𝗿𝘆 𝗱𝗶𝘀𝗲𝗮𝘀𝗲. […]
𝐒𝐲𝐧𝐞𝐫𝐠𝐢𝐬𝐭𝐢𝐜 𝐓𝐨𝐦𝐨𝐠𝐫𝐚𝐩𝐡𝐲: 𝐑𝐔𝐒-𝐏𝐀𝐓 𝐔𝐧𝐢𝐟𝐢𝐞𝐬 𝐒𝐭𝐫𝐮𝐜𝐭𝐮𝐫𝐚𝐥 𝐚𝐧𝐝 𝐅𝐮𝐧𝐜𝐭𝐢𝐨𝐧𝐚𝐥 𝐈𝐦𝐚𝐠𝐢𝐧𝐠 𝐢𝐧 𝐚 𝐒𝐢𝐧𝐠𝐥𝐞, 𝐑𝐚𝐩𝐢𝐝 𝐌𝐨𝐝𝐚𝐥𝐢𝐭𝐲 (Colored Ultrasound Imaging)
A significant advancement in medical imaging is detailed in the January 16 issue of Nature Biomedical Engineering. Researchers from Caltech and USC have developed Rotational Ultrasound and Photoacoustic Tomography (RUS-PAT), a novel hybrid technique that overcomes the fundamental limitations of standalone ultrasound and photoacoustic imaging to deliver rapid, co-registered 3D structural and functional data. The Core Innovation: Traditional […]
α-𝐒𝐲𝐧𝐮𝐜𝐥𝐞𝐢𝐧 𝐩𝐚𝐭𝐡𝐨𝐥𝐨𝐠𝐢𝐜𝐚𝐥 𝐚𝐠𝐠𝐫𝐞𝐠𝐚𝐭𝐢𝐨𝐧 𝐢𝐧𝐭𝐨 𝐢𝐧𝐬𝐨𝐥𝐮𝐛𝐥𝐞 𝐟𝐢𝐛𝐫𝐢𝐥𝐬 (𝐟𝐨𝐫𝐦𝐢𝐧𝐠 𝐋𝐞𝐰𝐲 𝐛𝐨𝐝𝐢𝐞𝐬)
𝐌𝐲 𝐫𝐞𝐬𝐞𝐚𝐫𝐜𝐡 𝐰𝐨𝐫𝐤 𝐨𝐧 𝐀𝐥𝐳𝐡𝐞𝐢𝐦𝐞𝐫’𝐬 𝐝𝐢𝐬𝐞𝐚𝐬𝐞 𝐭𝐨𝐝𝐚𝐲 𝐜𝐨𝐯𝐞𝐫𝐬 𝐦𝐚𝐧𝐲 𝐭𝐨𝐩𝐢𝐜𝐬 𝐢𝐧𝐜𝐥𝐮𝐝𝐢𝐧𝐠 α-𝐒𝐲𝐧𝐮𝐜𝐥𝐞𝐢𝐧, 𝐚 𝟏𝟒𝟎-𝐚𝐦𝐢𝐧𝐨 𝐚𝐜𝐢𝐝 𝐩𝐫𝐨𝐭𝐞𝐢𝐧 𝐩𝐫𝐢𝐦𝐚𝐫𝐢𝐥𝐲 𝐟𝐨𝐮𝐧𝐝 𝐢𝐧 𝐧𝐞𝐮𝐫𝐚𝐥 𝐭𝐢𝐬𝐬𝐮𝐞 (𝐬𝐩𝐞𝐜𝐢𝐟𝐢𝐜𝐚𝐥𝐥𝐲 𝐩𝐫𝐞𝐬𝐲𝐧𝐚𝐩𝐭𝐢𝐜 𝐭𝐞𝐫𝐦𝐢𝐧𝐚𝐥𝐬). α-𝐒𝐲𝐧𝐮𝐜𝐥𝐞𝐢𝐧 𝐩𝐥𝐚𝐲𝐬 𝐚 𝐤𝐞𝐲 𝐫𝐨𝐥𝐞 𝐢𝐧 𝐬𝐲𝐧𝐚𝐩𝐭𝐢𝐜 𝐯𝐞𝐬𝐢𝐜𝐥𝐞 𝐭𝐫𝐚𝐟𝐟𝐢𝐜𝐤𝐢𝐧𝐠, 𝐧𝐞𝐮𝐫𝐨𝐭𝐫𝐚𝐧𝐬𝐦𝐢𝐭𝐭𝐞𝐫 𝐫𝐞𝐥𝐞𝐚𝐬𝐞, 𝐚𝐧𝐝 𝐩𝐨𝐭𝐞𝐧𝐭𝐢𝐚𝐥𝐥𝐲 𝐧𝐞𝐮𝐫𝐨𝐧𝐚𝐥 𝐬𝐮𝐫𝐯𝐢𝐯𝐚𝐥. 𝐖𝐡𝐢𝐥𝐞 𝐢𝐭𝐬 𝐞𝐱𝐚𝐜𝐭 𝐩𝐡𝐲𝐬𝐢𝐨𝐥𝐨𝐠𝐢𝐜𝐚𝐥 𝐟𝐮𝐧𝐜𝐭𝐢𝐨𝐧 𝐫𝐞𝐦𝐚𝐢𝐧𝐬 𝐮𝐧𝐝𝐞𝐫 𝐢𝐧𝐯𝐞𝐬𝐭𝐢𝐠𝐚𝐭𝐢𝐨𝐧, 𝐢𝐭𝐬 𝐩𝐚𝐭𝐡𝐨𝐥𝐨𝐠𝐢𝐜𝐚𝐥 𝐚𝐠𝐠𝐫𝐞𝐠𝐚𝐭𝐢𝐨𝐧 𝐢𝐧𝐭𝐨 𝐢𝐧𝐬𝐨𝐥𝐮𝐛𝐥𝐞 𝐟𝐢𝐛𝐫𝐢𝐥𝐬 (𝐟𝐨𝐫𝐦𝐢𝐧𝐠 𝐋𝐞𝐰𝐲 […]



 Posted in
Posted in 