𝗡𝗜𝗖𝗘 𝗥𝗲𝗰𝗼𝗺𝗺𝗲𝗻𝗱𝘀 𝗗𝘂𝗽𝗶𝗹𝘂𝗺𝗮𝗯 𝗳𝗼𝗿 𝗘𝗼𝘀𝗶𝗻𝗼𝗽𝗵𝗶𝗹𝗶𝗰 𝗖𝗢𝗣𝗗: 𝗔 𝗗𝗲𝗲𝗽 𝗗𝗶𝘃𝗲 𝗶𝗻𝘁𝗼 𝘁𝗵𝗲 𝗘𝘃𝗶𝗱𝗲𝗻𝗰𝗲 𝗮𝗻𝗱 𝗘𝗰𝗼𝗻𝗼𝗺𝗶𝗰 𝗖𝗮𝘀𝗲
𝗧𝗵𝗲 𝗨𝗞’𝘀 𝗡𝗮𝘁𝗶𝗼𝗻𝗮𝗹 𝗜𝗻𝘀𝘁𝗶𝘁𝘂𝘁𝗲 𝗳𝗼𝗿 𝗛𝗲𝗮𝗹𝘁𝗵 𝗮𝗻𝗱 𝗖𝗮𝗿𝗲 𝗘𝘅𝗰𝗲𝗹𝗹𝗲𝗻𝗰𝗲 (𝗡𝗜𝗖𝗘) 𝗵𝗮𝘀 𝗶𝘀𝘀𝘂𝗲𝗱 𝗳𝗶𝗻𝗮𝗹 𝗱𝗿𝗮𝗳𝘁 𝗴𝘂𝗶𝗱𝗮𝗻𝗰𝗲 𝗿𝗲𝗰𝗼𝗺𝗺𝗲𝗻𝗱𝗶𝗻𝗴 𝗱𝘂𝗽𝗶𝗹𝘂𝗺𝗮𝗯 𝗮𝘀 𝗮𝗻 𝗮𝗱𝗱-𝗼𝗻 𝗺𝗮𝗶𝗻𝘁𝗲𝗻𝗮𝗻𝗰𝗲 𝘁𝗵𝗲𝗿𝗮𝗽𝘆 𝗳𝗼𝗿 𝗮 𝘀𝗽𝗲𝗰𝗶𝗳𝗶𝗰, 𝗵𝗶𝗴𝗵-𝗻𝗲𝗲𝗱 𝗖𝗢𝗣𝗗 𝗽𝗼𝗽𝘂𝗹𝗮𝘁𝗶𝗼𝗻. 𝗧𝗵𝗶𝘀 𝗺𝗮𝗿𝗸𝘀 𝗮 𝗽𝗶𝘃𝗼𝘁𝗮𝗹 𝘀𝗵𝗶𝗳𝘁 𝘁𝗼𝘄𝗮𝗿𝗱𝘀 𝗽𝗿𝗲𝗰𝗶𝘀𝗶𝗼𝗻 𝗺𝗲𝗱𝗶𝗰𝗶𝗻𝗲 𝗶𝗻 𝗰𝗵𝗿𝗼𝗻𝗶𝗰 𝗿𝗲𝘀𝗽𝗶𝗿𝗮𝘁𝗼𝗿𝘆 𝗱𝗶𝘀𝗲𝗮𝘀𝗲.
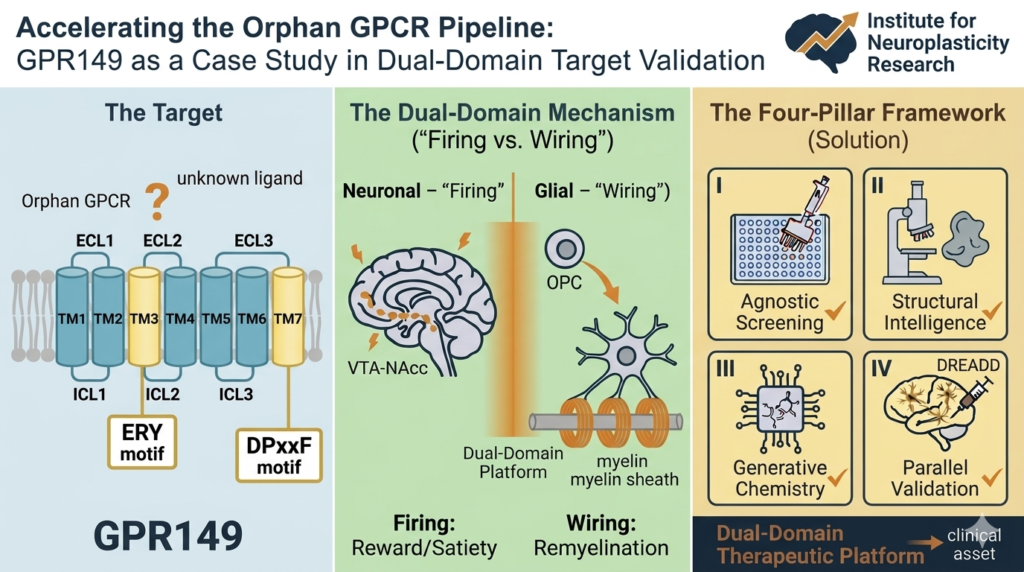
🏷️ Target Phenotype: Adults with uncontrolled COPD and raised blood eosinophils (≥300 cells/μL), despite being on maximal background therapy (triple or appropriate double therapy).
📊 Clinical Evidence (BOREAS & NOTUS Pooled Analysis):
31% reduction in annualized moderate/severe exacerbations (RR 0.69).
Significant lung function improvements: +83 mL in pre-bronchodilator FEV1 at week 12.
Meaningful quality-of-life gains (SGRQ score improvement).
💷 Cost-Effectiveness: The approval hinges on a robust health economic model. The committee-preferred ICER of £23,113 per QALY gained falls within NICE’s acceptable range, supported by a commercial access agreement. The model innovatively integrates:
Lifetime Markov structure with GOLD-stage-specific health states.
FEV1 decline rates adjusted for the eosinophilic phenotype.
Extrapolated long-term treatment effects from asthma data (TRAVERSE).
⚖️ Key Considerations & Implications:
Stopping Rule: Mandates 12-month response assessment, discontinuing if exacerbations do not meaningfully decrease.
Real-World Evidence Gap: Highlights the need for post-approval studies to confirm long-term outcomes and ICER stability.
Market Access Precedent: Successfully demonstrates value for a phenotype-specific biologic in COPD, potentially shaping future reimbursement negotiations in single-payer systems.
This decision is more than a new therapy; it’s a blueprint for integrating advanced biologics into COPD management, demanding careful patient selection, monitoring, and ongoing evidence generation.
hashtag#COPD hashtag#Dupilumab hashtag#EosinophilicCOPD hashtag#NICE hashtag#HealthEconomics hashtag#MarketAccess hashtag#HEOR hashtag#PrecisionMedicine hashtag#Biologics hashtag#RespiratoryMedicine
𝐀 𝐍𝐞𝐰 𝐂𝐡𝐚𝐩𝐭𝐞𝐫 𝐟𝐨𝐫 𝐒𝐞𝐯𝐞𝐫𝐞 𝐂𝐎𝐏𝐃 𝐂𝐚𝐫𝐞: 𝐍𝐇𝐒 𝐭𝐨 𝐅𝐮𝐧𝐝 𝐅𝐢𝐫𝐬𝐭 𝐓𝐚𝐫𝐠𝐞𝐭𝐞𝐝 𝐁𝐢𝐨𝐥𝐨𝐠𝐢𝐜
𝐁𝐫𝐞𝐚𝐤𝐢𝐧𝐠 𝐧𝐞𝐰𝐬 𝐟𝐨𝐫 𝐭𝐡𝐞 𝐫𝐞𝐬𝐩𝐢𝐫𝐚𝐭𝐨𝐫𝐲 𝐜𝐨𝐦𝐦𝐮𝐧𝐢𝐭𝐲: 𝐍𝐈𝐂𝐄 𝐡𝐚𝐬 𝐠𝐫𝐞𝐞𝐧𝐥𝐢𝐭 𝐝𝐮𝐩𝐢𝐥𝐮𝐦𝐚𝐛 𝐟𝐨𝐫 𝐍𝐇𝐒 𝐮𝐬𝐞 𝐢𝐧 𝐚 𝐬𝐩𝐞𝐜𝐢𝐟𝐢𝐜 𝐠𝐫𝐨𝐮𝐩 𝐨𝐟 𝐩𝐚𝐭𝐢𝐞𝐧𝐭𝐬 𝐰𝐢𝐭𝐡 𝐬𝐞𝐯𝐞𝐫𝐞 𝐂𝐎𝐏𝐃. 𝐓𝐡𝐢𝐬 𝐢𝐬 𝐭𝐡𝐞 𝐟𝐢𝐫𝐬𝐭 𝐞𝐨𝐬𝐢𝐧𝐨𝐩𝐡𝐢𝐥-𝐭𝐚𝐫𝐠𝐞𝐭𝐞𝐝 𝐛𝐢𝐨𝐥𝐨𝐠𝐢𝐜 𝐭𝐫𝐞𝐚𝐭𝐦𝐞𝐧𝐭 𝐟𝐨𝐫 𝐂𝐎𝐏𝐃 𝐢𝐧 𝐭𝐡𝐞 𝐔𝐊, 𝐨𝐟𝐟𝐞𝐫𝐢𝐧𝐠 𝐧𝐞𝐰 𝐡𝐨𝐩𝐞 𝐟𝐨𝐫 𝐚 𝐝𝐢𝐟𝐟𝐢𝐜𝐮𝐥𝐭-𝐭𝐨-𝐭𝐫𝐞𝐚𝐭 𝐜𝐨𝐧𝐝𝐢𝐭𝐢𝐨𝐧.
𝐖𝐡𝐨 𝐜𝐨𝐮𝐥𝐝 𝐛𝐞𝐧𝐞𝐟𝐢𝐭? 𝐀𝐝𝐮𝐥𝐭𝐬 𝐰𝐢𝐭𝐡 𝐚 𝐬𝐩𝐞𝐜𝐢𝐟𝐢𝐜 𝐭𝐲𝐩𝐞 𝐨𝐟 𝐂𝐎𝐏𝐃 𝐦𝐚𝐫𝐤𝐞𝐝 𝐛𝐲 𝐡𝐢𝐠𝐡 𝐥𝐞𝐯𝐞𝐥𝐬 𝐨𝐟 𝐚 𝐜𝐞𝐫𝐭𝐚𝐢𝐧 𝐰𝐡𝐢𝐭𝐞 𝐛𝐥𝐨𝐨𝐝 𝐜𝐞𝐥𝐥 (𝐞𝐨𝐬𝐢𝐧𝐨𝐩𝐡𝐢𝐥𝐬), 𝐰𝐡𝐨 𝐜𝐨𝐧𝐭𝐢𝐧𝐮𝐞 𝐭𝐨 𝐬𝐮𝐟𝐟𝐞𝐫 𝐬𝐞𝐫𝐢𝐨𝐮𝐬 𝐟𝐥𝐚𝐫𝐞-𝐮𝐩𝐬 (“𝐞𝐱𝐚𝐜𝐞𝐫𝐛𝐚𝐭𝐢𝐨𝐧𝐬”) 𝐝𝐞𝐬𝐩𝐢𝐭𝐞 𝐮𝐬𝐢𝐧𝐠 𝐬𝐭𝐚𝐧𝐝𝐚𝐫𝐝 𝐢𝐧𝐡𝐚𝐥𝐞𝐫 𝐭𝐡𝐞𝐫𝐚𝐩𝐢𝐞𝐬.
𝐖𝐡𝐲 𝐢𝐬 𝐭𝐡𝐢𝐬 𝐬𝐢𝐠𝐧𝐢𝐟𝐢𝐜𝐚𝐧𝐭?
𝐓𝐚𝐫𝐠𝐞𝐭𝐞𝐝 𝐀𝐩𝐩𝐫𝐨𝐚𝐜𝐡: 𝐌𝐨𝐯𝐞𝐬 𝐛𝐞𝐲𝐨𝐧𝐝 “𝐨𝐧𝐞-𝐬𝐢𝐳𝐞-𝐟𝐢𝐭𝐬-𝐚𝐥𝐥” 𝐭𝐫𝐞𝐚𝐭𝐦𝐞𝐧𝐭 𝐭𝐨 𝐭𝐚𝐫𝐠𝐞𝐭 𝐚𝐧 𝐮𝐧𝐝𝐞𝐫𝐥𝐲𝐢𝐧𝐠 𝐝𝐫𝐢𝐯𝐞𝐫 𝐨𝐟 𝐢𝐧𝐟𝐥𝐚𝐦𝐦𝐚𝐭𝐢𝐨𝐧 𝐢𝐧 𝐚 𝐝𝐞𝐟𝐢𝐧𝐞𝐝 𝐩𝐚𝐭𝐢𝐞𝐧𝐭 𝐬𝐮𝐛𝐠𝐫𝐨𝐮𝐩.
𝐏𝐫𝐨𝐯𝐞𝐧 𝐎𝐮𝐭𝐜𝐨𝐦𝐞𝐬: 𝐈𝐧 𝐜𝐥𝐢𝐧𝐢𝐜𝐚𝐥 𝐭𝐫𝐢𝐚𝐥𝐬, 𝐢𝐭 𝐜𝐮𝐭 𝐭𝐡𝐞 𝐫𝐚𝐭𝐞 𝐨𝐟 𝐝𝐞𝐛𝐢𝐥𝐢𝐭𝐚𝐭𝐢𝐧𝐠 𝐟𝐥𝐚𝐫𝐞-𝐮𝐩𝐬 𝐛𝐲 𝐧𝐞𝐚𝐫𝐥𝐲 𝐨𝐧𝐞-𝐭𝐡𝐢𝐫𝐝 𝐚𝐧𝐝 𝐡𝐞𝐥𝐩𝐞𝐝 𝐢𝐦𝐩𝐫𝐨𝐯𝐞 𝐥𝐮𝐧𝐠 𝐟𝐮𝐧𝐜𝐭𝐢𝐨𝐧 𝐚𝐧𝐝 𝐪𝐮𝐚𝐥𝐢𝐭𝐲 𝐨𝐟 𝐥𝐢𝐟𝐞.
𝐀𝐝𝐝𝐫𝐞𝐬𝐬𝐞𝐬 𝐚 𝐇𝐢𝐠𝐡 𝐁𝐮𝐫𝐝𝐞𝐧: 𝐂𝐎𝐏𝐃 𝐝𝐢𝐬𝐩𝐫𝐨𝐩𝐨𝐫𝐭𝐢𝐨𝐧𝐚𝐭𝐞𝐥𝐲 𝐚𝐟𝐟𝐞𝐜𝐭𝐬 𝐩𝐞𝐨𝐩𝐥𝐞 𝐢𝐧 𝐦𝐨𝐫𝐞 𝐝𝐞𝐩𝐫𝐢𝐯𝐞𝐝 𝐚𝐫𝐞𝐚𝐬. 𝐓𝐡𝐢𝐬 𝐧𝐞𝐰 𝐨𝐩𝐭𝐢𝐨𝐧 𝐚𝐢𝐦𝐬 𝐭𝐨 𝐫𝐞𝐝𝐮𝐜𝐞 𝐡𝐨𝐬𝐩𝐢𝐭𝐚𝐥𝐢𝐳𝐚𝐭𝐢𝐨𝐧𝐬 𝐚𝐧𝐝 𝐡𝐞𝐚𝐥𝐭𝐡 𝐢𝐧𝐞𝐪𝐮𝐚𝐥𝐢𝐭𝐢𝐞𝐬.
𝐂𝐨𝐬𝐭-𝐄𝐟𝐟𝐞𝐜𝐭𝐢𝐯𝐞 𝐈𝐧𝐧𝐨𝐯𝐚𝐭𝐢𝐨𝐧: 𝐁𝐚𝐜𝐤𝐞𝐝 𝐛𝐲 𝐚 𝐩𝐨𝐬𝐢𝐭𝐢𝐯𝐞 𝐜𝐨𝐬𝐭-𝐞𝐟𝐟𝐞𝐜𝐭𝐢𝐯𝐞𝐧𝐞𝐬𝐬 𝐚𝐬𝐬𝐞𝐬𝐬𝐦𝐞𝐧𝐭 𝐚𝐧𝐝 𝐚 𝐜𝐨𝐦𝐦𝐞𝐫𝐜𝐢𝐚𝐥 𝐚𝐠𝐫𝐞𝐞𝐦𝐞𝐧𝐭, 𝐞𝐧𝐬𝐮𝐫𝐢𝐧𝐠 𝐯𝐚𝐥𝐮𝐞 𝐟𝐨𝐫 𝐭𝐡𝐞 𝐍𝐇𝐒.
The Bottom Line: This isn’t just a new drug. It represents a major step forward in personalising COPD care. It validates the importance of identifying specific patient phenotypes and paves the way for more targeted therapies in respiratory disease. For eligible patients, it promises fewer flare-ups, better breathing, and a chance at a more stable life.
The NHS must now implement this within 90 days, highlighting the rapid translation of evidence into practice for patient benefit.
hashtag#HealthcareInnovation hashtag#NHS hashtag#RespiratoryHealth hashtag#COPDAwareness hashtag#PatientCare hashtag#Pharma hashtag#Biotech hashtag#PublicHealth hashtag#UKHealthcare